|
This table shows how the atom size, and atomic radius values change as you move horizontally and vertically across the periodic table. 
Pb State the charge that each of the following would have if it becomes an ion: a. He Which of the following atoms has the most metallic character a. As you know, atomic radii decreases across a Period, from left to right as we face the Table, and increases down a Group. P Which of the following atoms has the lowest first ionization energy a. Therefore, the radius of an atom is more than 10,000 times the radius of its nucleus (110 fm ), 2 and less than 1/1000 of the wavelength of visible light (400700 nm ). Which of the following atoms has the largest atomic radius a. What is the effective nuclear charge (Z eff) of valence electron on Mg 10, 12, 3, 1, or 2 5. Under most definitions the radii of isolated neutral atoms range between 30 and 300 pm ( trillionths of a meter), or between 0.3 and 3 ngstrms. Which element has the most non-metallic character Sr Te Ga Cl Be 4. 
Which element has the most metallic character Br S Si Li Fr 3. Atom size values are calculated from atomic radius data. Which of the following atoms has the largest atomic radius Ge Rb S Br Na 2. 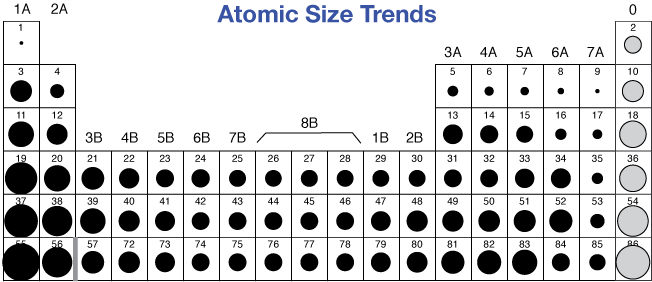
Each atom’s size is relative to the largest element, cesium. (g) e^- \nonumber \]Įach successive IE is larger than the previous because an electron is being removed from an atom with a progressively larger positive charge. This periodic table shows the relative sizes of the atoms of each element. As you go down a column of the periodic table, the atomic radii increase.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
